Recyclable high-strength polybutadiene-based rubber with self-healing and shape memory properties via dynamic boronic ester and Diels-Alder chemistry
Shengao Yang, Yan Wang, Fang Wang, Kaiyi Zhang, Xinxin Lv, Hao Teng, Rui Zheng, Faliang Luo, Qian Xing
Vol. 19., No.1., Pages 94-106, 2025
DOI: 10.3144/expresspolymlett.2025.7
DOI: 10.3144/expresspolymlett.2025.7
GRAPHICAL ABSTRACT
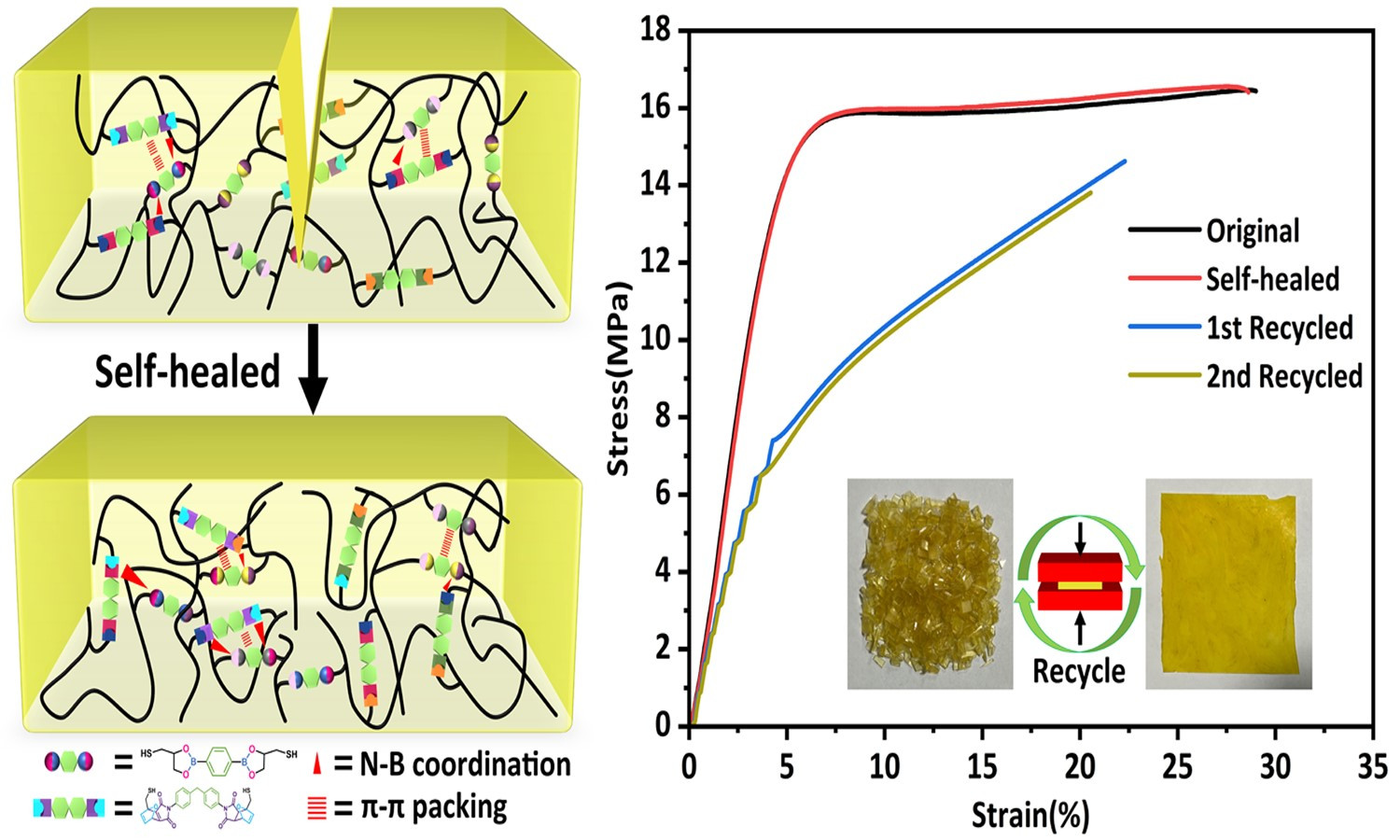
ABSTRACT
Dynamic cross-linked networks (DCNs) endow thermoset rubber with self-healability and recyclability to extend its lifetime and alleviate environmental pollution. However, the contradiction between high self-healing and mechanical properties in DCNs rubber is always difficult to be resolved. Herein, we used boronic ester (BO) and Diels-Alder dynamic covalent bonds (DA) to synthesize polybutadiene-based dual networks rubber (PB-BO-DA) via thiol-ene reaction. This approach achieved a tensile strength of 16.46 MPa and 99% self-healing efficiency, facilitated by extensive intermolecular interactions (π-π packing and N-B coordination) and fully dynamic cross-linking. In addition, multiple dynamic cross-linked networks (MDCNs) polybutadiene-based rubber also show excellent shape memory ability and recyclability. This strategy might open a helpful pathway to fabricate intelligent multifunctional polymers with high strength and high self-healing efficiency.
RELATED ARTICLES
Wu Qin, Rui Zhang, Long Zheng, Danling Wang, Sheng Chen, Huiming Ren, Shui Hu, Shipeng Wen
Vol. 20., No.6., Pages 551-562, 2026
DOI: 10.3144/expresspolymlett.2026.42
Vol. 20., No.6., Pages 551-562, 2026
DOI: 10.3144/expresspolymlett.2026.42

Conventional sulfur vulcanization in rubber manufacturing depends on elevated curing temperatures and zinc oxide activators, resulting in high energy consumption and increasing environmental concerns associated with zinc release. To overcome these limitations, a novel graphene oxide (GO)-supported rare-earth-containing accelerator (GO–LZC) was designed by coordinating lanthanum(III) and zinc(II) ions with sodium diethyldithiocarbamate (DC). At the same time, the oxygen-containing groups on GO further participated in ligand coordination. The resulting GO-immobilized complex exhibits a well-defined chelating structure and uniform nanoscale dispersion, which together enhance the accessibility and reactivity of active sulfurating species during curing. When incorporated into solution-polymerized styrene–butadiene rubber (SSBR), GO–LZC markedly promotes crosslink formation at reduced thermal input. Kinetic analysis reveals a substantial decrease in the apparent activation energy, and curing and mechanical tests confirm that efficient vulcanization can be achieved at 130 °C, representing a 20–40 °C reduction relative to typical industrial curing conditions. This work demonstrates a viable strategy for developing low-zinc, energy-efficient, and high-performance vulcanization systems. It highlights the potential of rare-earth/GO hybrid catalysts for sustainable rubber processing.
Using dual-cure architectures in HNBR: A detailed insight into their structure-property relationship
Sharmistha Dhar, Arshad Rahman Parathodika, Dibyendu Dey, Kinsuk Naskar
Vol. 20., No.5., Pages 514-530, 2026
DOI: 10.3144/expresspolymlett.2026.39
Vol. 20., No.5., Pages 514-530, 2026
DOI: 10.3144/expresspolymlett.2026.39

Hydrogenated acrylonitrile–butadiene rubber (HNBR) is widely used in automotive and sealing applications due to its oil resistance and mechanical durability; however, its long-term performance is significantly influenced by the curing chemistry. Sulfur vulcanization offers superior elasticity but restricted thermal stability, while peroxide curing improves heat resistance at the expense of flexibility. In this study, we investigate hybrid sulfur–peroxide curing to integrate these benefits. The hybrid pathway encompasses competitive and sequential processes, such as partial radical quenching and accelerator oxidation, resulting in a dual crosslink network. Dynamic mechanical, thermal, and temperature scanning stress relaxation (TSSR) evaluations demonstrate that hybrid systems provide precise modulation of the operational temperature–frequency range, broaden the glass-transition relaxation, and control stress dissipation. The coexistence of sulfur and C–C crosslinks results in a heterogeneous structure characterized by diverse crosslink densities and bond energies, leading to numerous relaxation modes and an optimal blend of elasticity, strength, and thermal stability. Microscopy confirms the absence of phase separation, indicating that hybrid vulcanization is a viable approach for producing robust, high-performance HNBR elastomers.
Dibyendu Dey, Sharmistha Dhar, Barkat Aziz, Sambhu Bhadra, Sujith Nair, Kinsuk Naskar
Vol. 20., No.2., Pages 127-141, 2026
DOI: 10.3144/expresspolymlett.2026.11
Vol. 20., No.2., Pages 127-141, 2026
DOI: 10.3144/expresspolymlett.2026.11
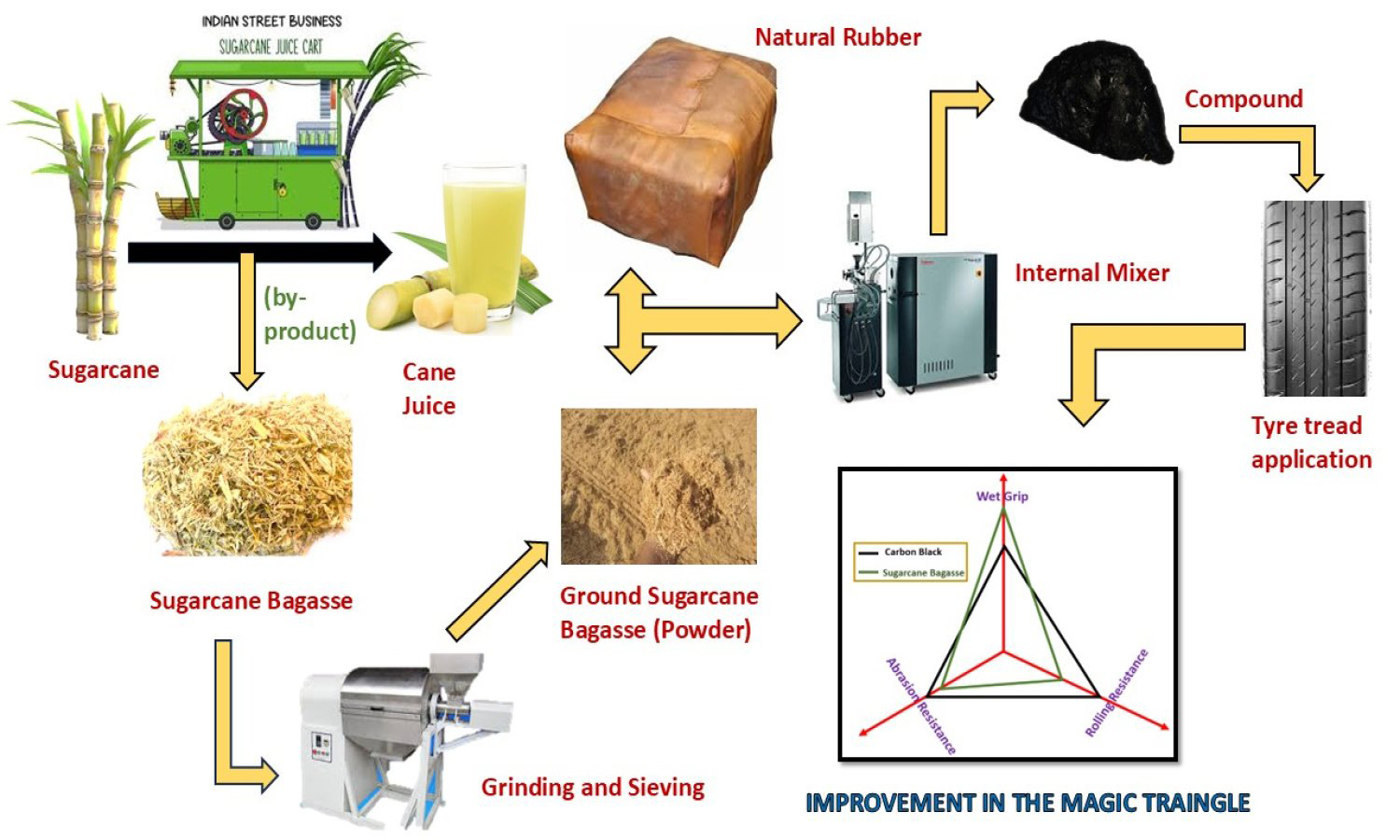
Ground sugarcane bagasse (GSB), an agro-waste rich in lignocellulosic components, was studied as a sustainable bio-filler in natural rubber (NR) tread compounds to lessen reliance on petroleum-derived carbon black (CB). A control formulation with 45 phr CB was compared to hybrid formulations with 40, 35, and 30 phr CB mixed with 5, 10, and 15 phr GSB. Tensile strength 13.1 MPa, elongation at break 700%, and hardness 67 Shore A were all optimally balanced by the compound containing 10 phr GSB (S2), while also exhibiting good cure behavior and thermal stability. Improved tire performance characteristics were confirmed by a dynamic mechanical study, which showed that tan δ at 60 °C decreased by 8.0% (resulting in lower rolling resistance) and increased by 3.9% (improving wet traction) at 0°C. The Payne effect showed improved filler dispersion as a result of GSB partially replacing CB. The results show that appropriately dispersed GSB can partially reinforce NR, enhancing energy efficiency and sustainability. However, larger GSB loadings decrease modulus, tear strength, and abrasion resistance due to lower interfacial adhesion and the presence of micro-voids. According to this study, pulverized sugarcane bagasse shows promise as an environmentally friendly filler for green tire applications, promoting the circular economy and lowering the carbon footprint of rubber compounding.
Rattanawadee Ninjan, Bencha Thongnuanchan, Phakawat Tongnuanchan, Subhan Salaeh, Jutharat Intapun, Abdulhakim Masa, Natinee Lopattananon
Vol. 20., No.1., Pages 18-35, 2026
DOI: 10.3144/expresspolymlett.2026.3
Vol. 20., No.1., Pages 18-35, 2026
DOI: 10.3144/expresspolymlett.2026.3
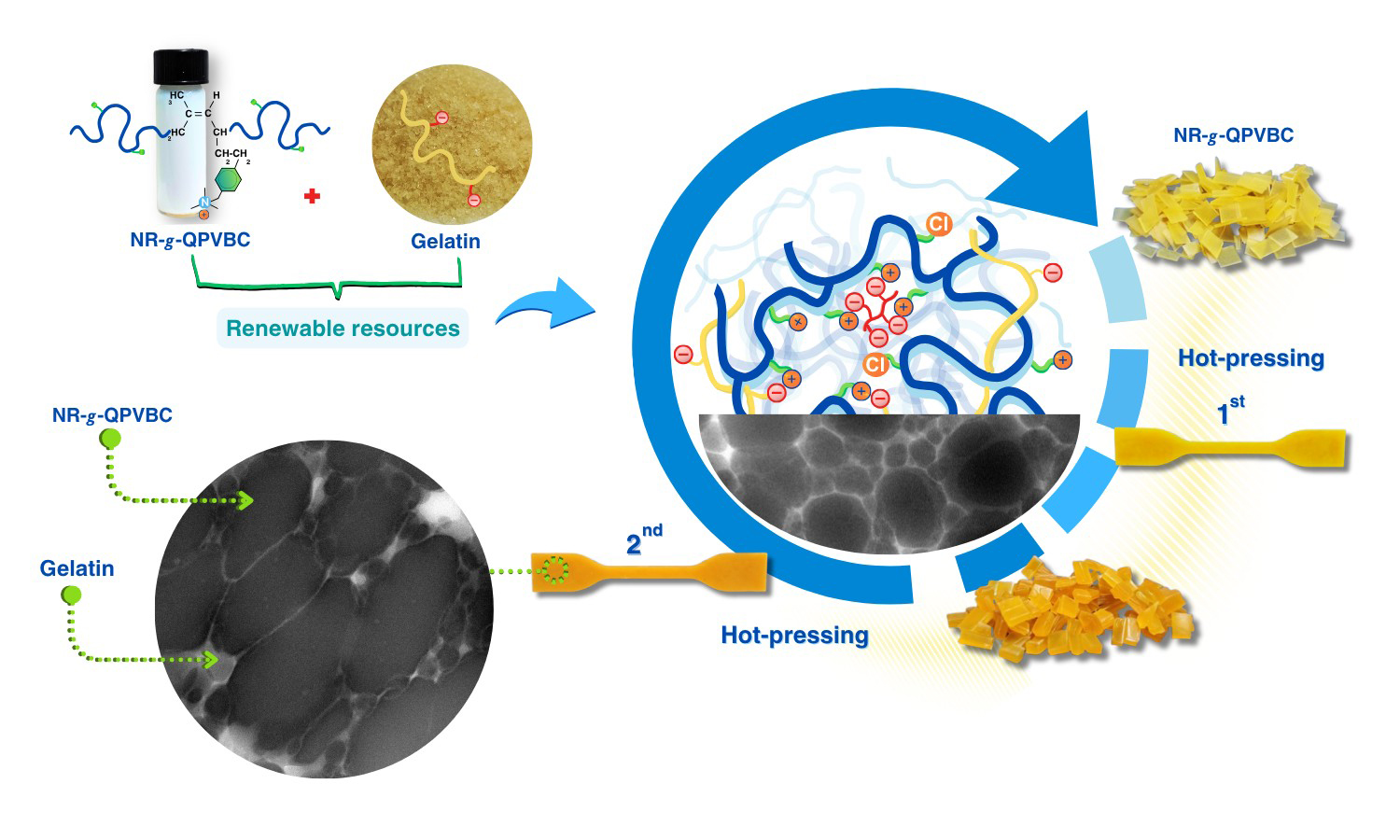
The present study has proposed a straightforward method to improve the reprocessability of modified natural rubber (NR) by blending it with gelatin (GT). The reprocessable characteristics of these blends were evaluated based on their remolding capabilities and mechanical recovery performance. In this method, poly(vinylbenzyl chloride) (PVBC) was first grafted onto NR chains to create graft copolymers known as NR-g-PVBC. The benzyl chloride groups in the graft copolymers were subsequently converted into quaternary ammonium groups, referred to as NR-g-QPVBC. This modification enabled ionic crosslinking when NR-g-QPVBC reacted with ethylenediamine tetraacetic acid. Blends were created by incorporating GT powder into the NR-g-QPVBC latex. The optimal loading level of GT was determined to be 30 wt%, as the resulting film exhibited the highest recovery of tensile properties. Initially, the film's tensile strength was measured at 15 MPa. After being remolded at 160 °C, the tensile strength decreased to 9.3 MPa, resulting in a recovery rate of 60.7% and withstanding a tensile strain of 144%. Although the NR-g-QPVBC/GT films could be remolded, their tensile properties declined with increasing remolding cycles. Therefore, this work demonstrated a practical method for producing NR-based films that could be reshaped through hot-pressing after being formed into products, increasing their reusability.
Cristian Valdés, Valentina Guzmán, Camila Ponce, Maribel Mamani, Juan Guevara, Claudia Vergara, Rodrigo Andler
Vol. 19., No.6., Pages 594-609, 2025
DOI: 10.3144/expresspolymlett.2025.45
Vol. 19., No.6., Pages 594-609, 2025
DOI: 10.3144/expresspolymlett.2025.45
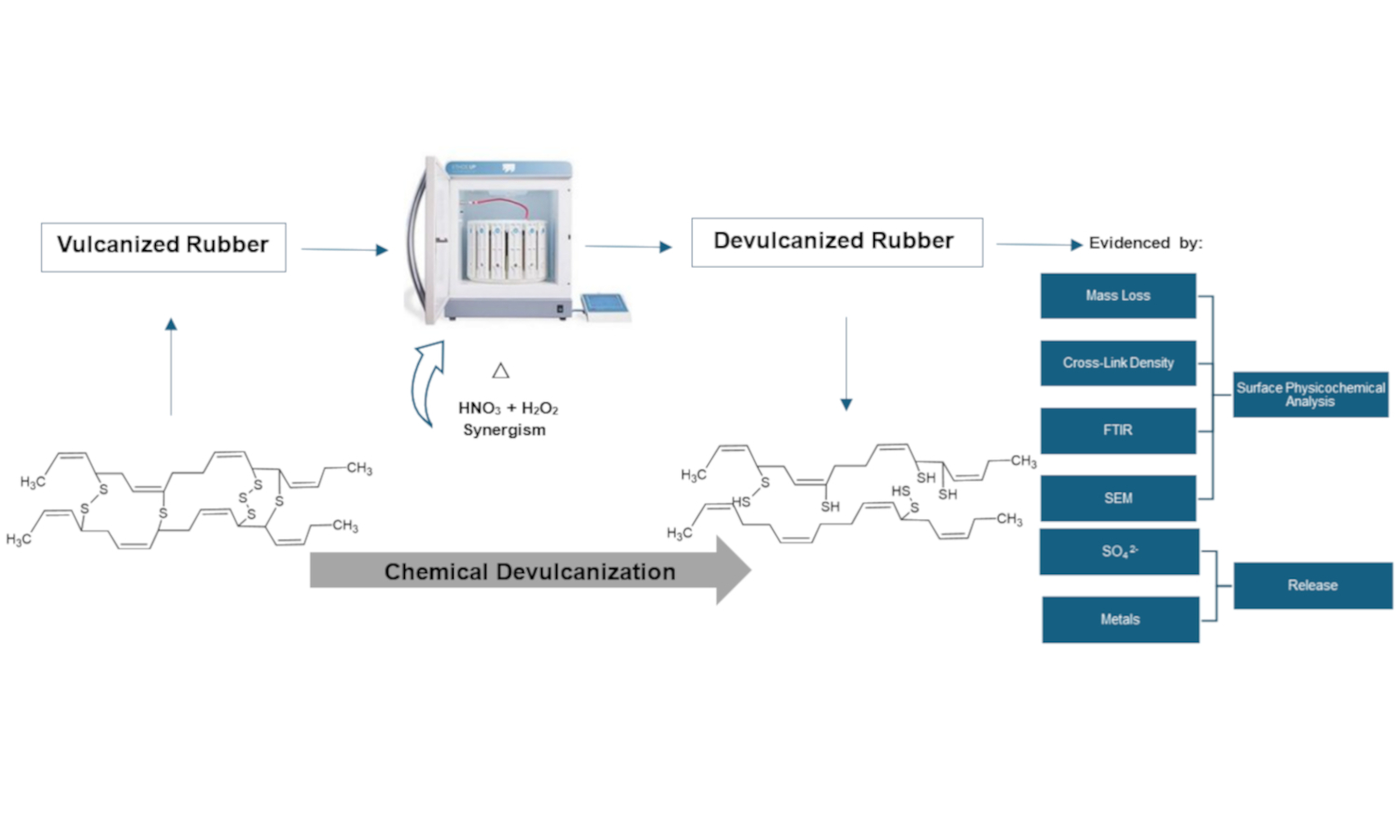
Waste rubber disposal causes considerable negative environmental impacts due to its increase worldwide, mainly in the automotive industry. Therefore, the search for technological solutions for rubber waste is a priority, and the first step in this material degradation is devulcanization due to its difficult degradation. This study evaluated rubber devulcanization using a closed vessel microwave digestion system with nitric acid (HNO3) and hydrogen peroxide (H2O2) through chemical characterization, aiming at verifying the synergistic effect between these oxidizing agents. Microwave irradiation was applied as a heating method to facilitate the chemical reactions, focusing on the synergism between HNO3 and H2O2. Results showed that 5 M H2O2 in combination with 1% HNO3, presented better results. A greater decrease in cross-link density was demonstrated as the concentration of H2O2 increased (3.96·10–5±1.99·10–6 mol/cm3), likewise, higher sulfates released (926.8±53.4 mg/L), increased mass loss (12.184±1.06%), rubber surface fragmentation, and important variations in the C–S, C=O bands, showing better results when devulcanization is carried out in synergism between HNO3 and H2O2.


