Coordination crosslinks of epoxidized natural rubber with reactive zinc chloride
Vol. 18., No.11., Pages 1149-1163, 2024
DOI: 10.3144/expresspolymlett.2024.87
DOI: 10.3144/expresspolymlett.2024.87
GRAPHICAL ABSTRACT
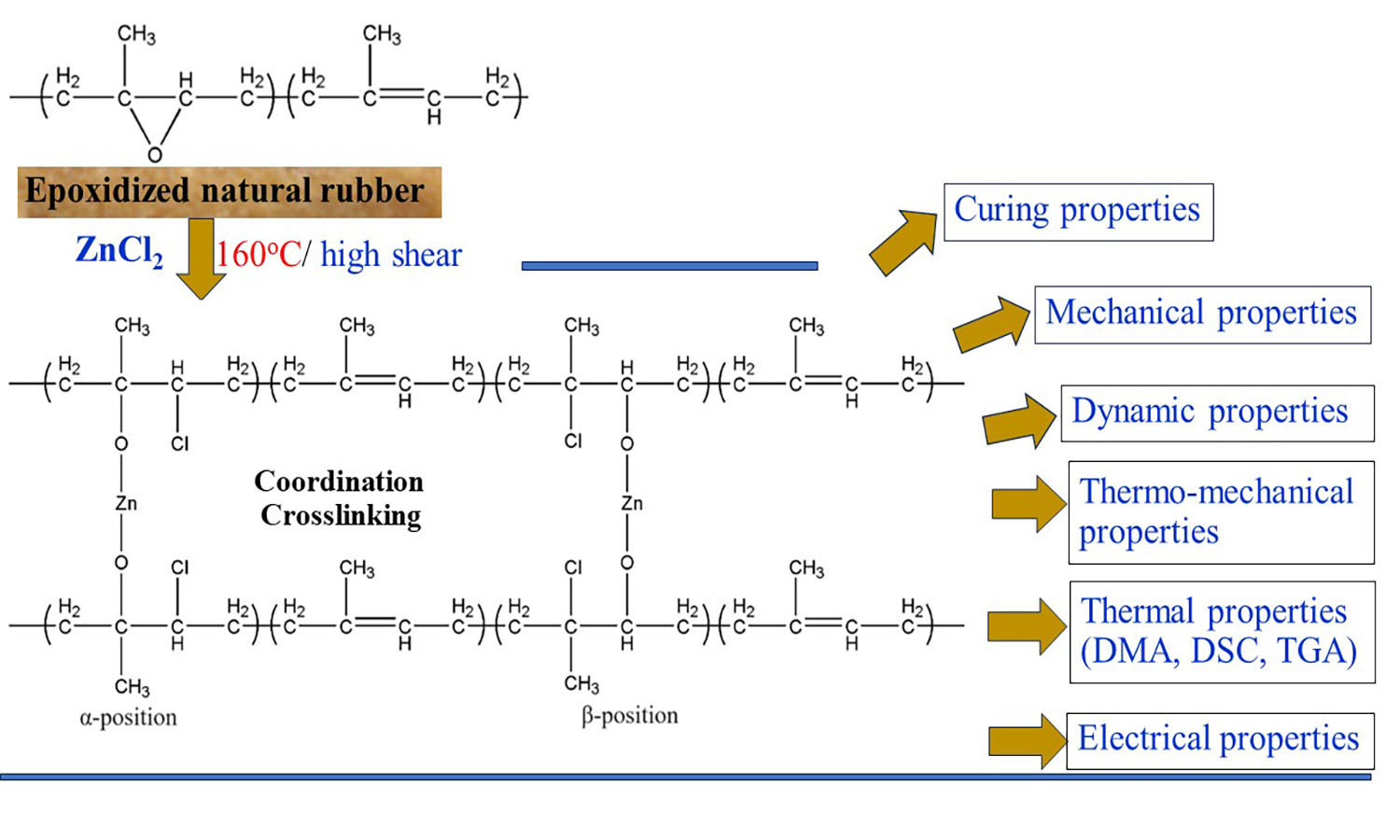
ABSTRACT
Epoxidized natural rubber with 50 mol% epoxide (ENR-50) was compounded with zinc chloride (ZnCl2) and subjected to torque response analysis using a moving die rheometer. It was found that different ZnCl2 concentrations (3, 5, 7, 9, and 12 millimoles (mmol)) mixed in ENR-50 exhibited positive torque responses, prompting further molecular characterization using Fourier transform infrared spectroscopy. The results indicated distinct absorption peaks at wavenumbers of 442 and 809 cm–1, which signify the presence of –O–Zn–O– coordination linkages. The curing characteristics of ENR and ZnCl2 compounds showed that increasing ZnCl2 content resulted in higher minimum and maximum torque values, but also led to a decrease in scorch time and cure rate index (CRI). Moreover, higher ZnCl2 concentrations enhanced the strength properties (tensile strength, moduli, stiffness, toughness, and hardness), crosslink densities, dynamic shear moduli, initial modulus during relaxation experiments, and thermal resistance, as evidenced by temperature scanning stress relaxation (TSSR), thermogravimetric analysis, and dynamic mechanical analysis. However, an increase in ZnCl2 content led to a reduction in elongation at break due to the higher crosslink density within the coordination networks in the ENR matrix, which resulted in the movement constraint of the rubber vulcanizate.
RELATED ARTICLES
Using dual-cure architectures in HNBR: A detailed insight into their structure-property relationship
Sharmistha Dhar, Arshad Rahman Parathodika, Dibyendu Dey, Kinsuk Naskar
Vol. 20., No.5., Pages 514-530, 2026
DOI: 10.3144/expresspolymlett.2026.39
Vol. 20., No.5., Pages 514-530, 2026
DOI: 10.3144/expresspolymlett.2026.39

Hydrogenated acrylonitrile–butadiene rubber (HNBR) is widely used in automotive and sealing applications due to its oil resistance and mechanical durability; however, its long-term performance is significantly influenced by the curing chemistry. Sulfur vulcanization offers superior elasticity but restricted thermal stability, while peroxide curing improves heat resistance at the expense of flexibility. In this study, we investigate hybrid sulfur–peroxide curing to integrate these benefits. The hybrid pathway encompasses competitive and sequential processes, such as partial radical quenching and accelerator oxidation, resulting in a dual crosslink network. Dynamic mechanical, thermal, and temperature scanning stress relaxation (TSSR) evaluations demonstrate that hybrid systems provide precise modulation of the operational temperature–frequency range, broaden the glass-transition relaxation, and control stress dissipation. The coexistence of sulfur and C–C crosslinks results in a heterogeneous structure characterized by diverse crosslink densities and bond energies, leading to numerous relaxation modes and an optimal blend of elasticity, strength, and thermal stability. Microscopy confirms the absence of phase separation, indicating that hybrid vulcanization is a viable approach for producing robust, high-performance HNBR elastomers.
Dibyendu Dey, Sharmistha Dhar, Barkat Aziz, Sambhu Bhadra, Sujith Nair, Kinsuk Naskar
Vol. 20., No.2., Pages 127-141, 2026
DOI: 10.3144/expresspolymlett.2026.11
Vol. 20., No.2., Pages 127-141, 2026
DOI: 10.3144/expresspolymlett.2026.11
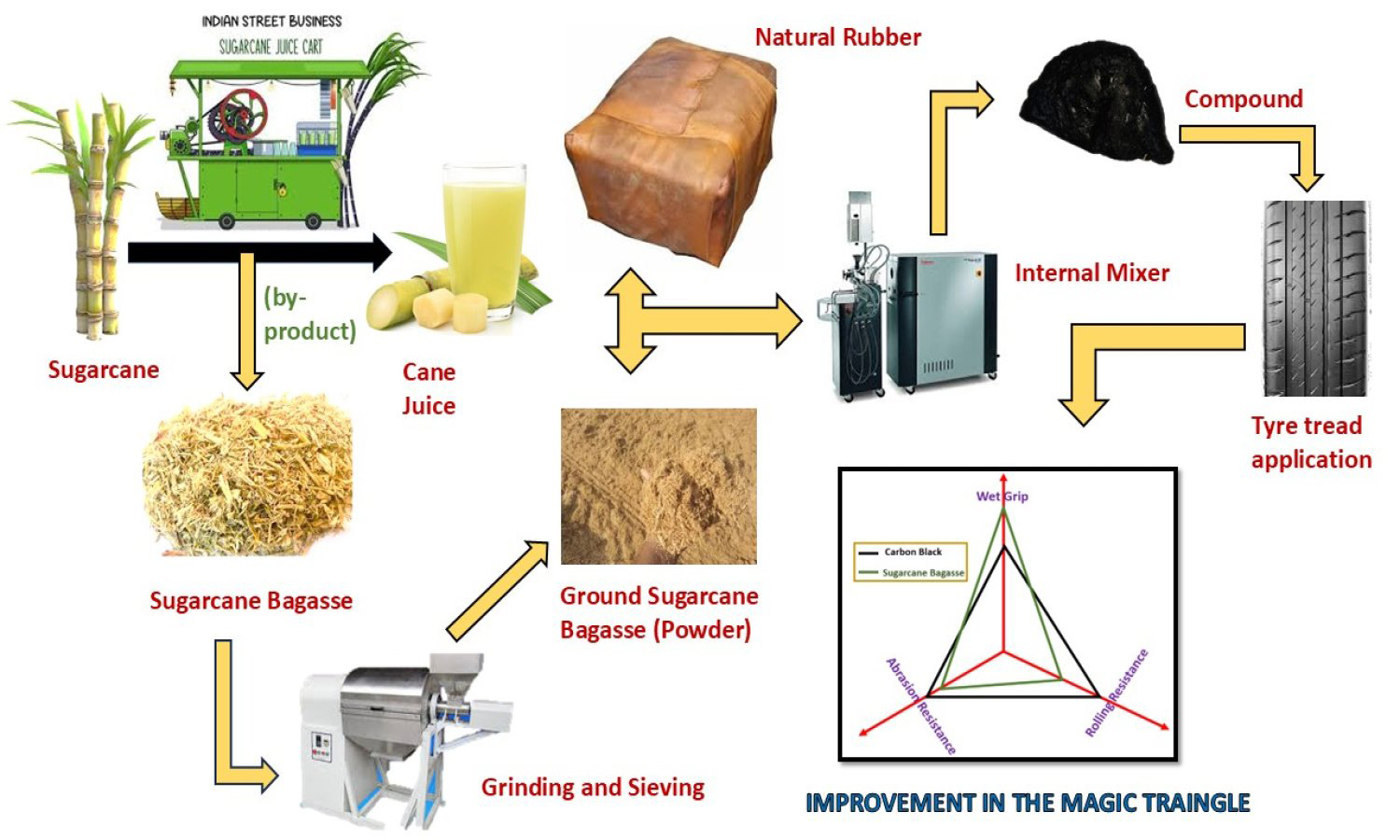
Ground sugarcane bagasse (GSB), an agro-waste rich in lignocellulosic components, was studied as a sustainable bio-filler in natural rubber (NR) tread compounds to lessen reliance on petroleum-derived carbon black (CB). A control formulation with 45 phr CB was compared to hybrid formulations with 40, 35, and 30 phr CB mixed with 5, 10, and 15 phr GSB. Tensile strength 13.1 MPa, elongation at break 700%, and hardness 67 Shore A were all optimally balanced by the compound containing 10 phr GSB (S2), while also exhibiting good cure behavior and thermal stability. Improved tire performance characteristics were confirmed by a dynamic mechanical study, which showed that tan δ at 60 °C decreased by 8.0% (resulting in lower rolling resistance) and increased by 3.9% (improving wet traction) at 0°C. The Payne effect showed improved filler dispersion as a result of GSB partially replacing CB. The results show that appropriately dispersed GSB can partially reinforce NR, enhancing energy efficiency and sustainability. However, larger GSB loadings decrease modulus, tear strength, and abrasion resistance due to lower interfacial adhesion and the presence of micro-voids. According to this study, pulverized sugarcane bagasse shows promise as an environmentally friendly filler for green tire applications, promoting the circular economy and lowering the carbon footprint of rubber compounding.
Rattanawadee Ninjan, Bencha Thongnuanchan, Phakawat Tongnuanchan, Subhan Salaeh, Jutharat Intapun, Abdulhakim Masa, Natinee Lopattananon
Vol. 20., No.1., Pages 18-35, 2026
DOI: 10.3144/expresspolymlett.2026.3
Vol. 20., No.1., Pages 18-35, 2026
DOI: 10.3144/expresspolymlett.2026.3
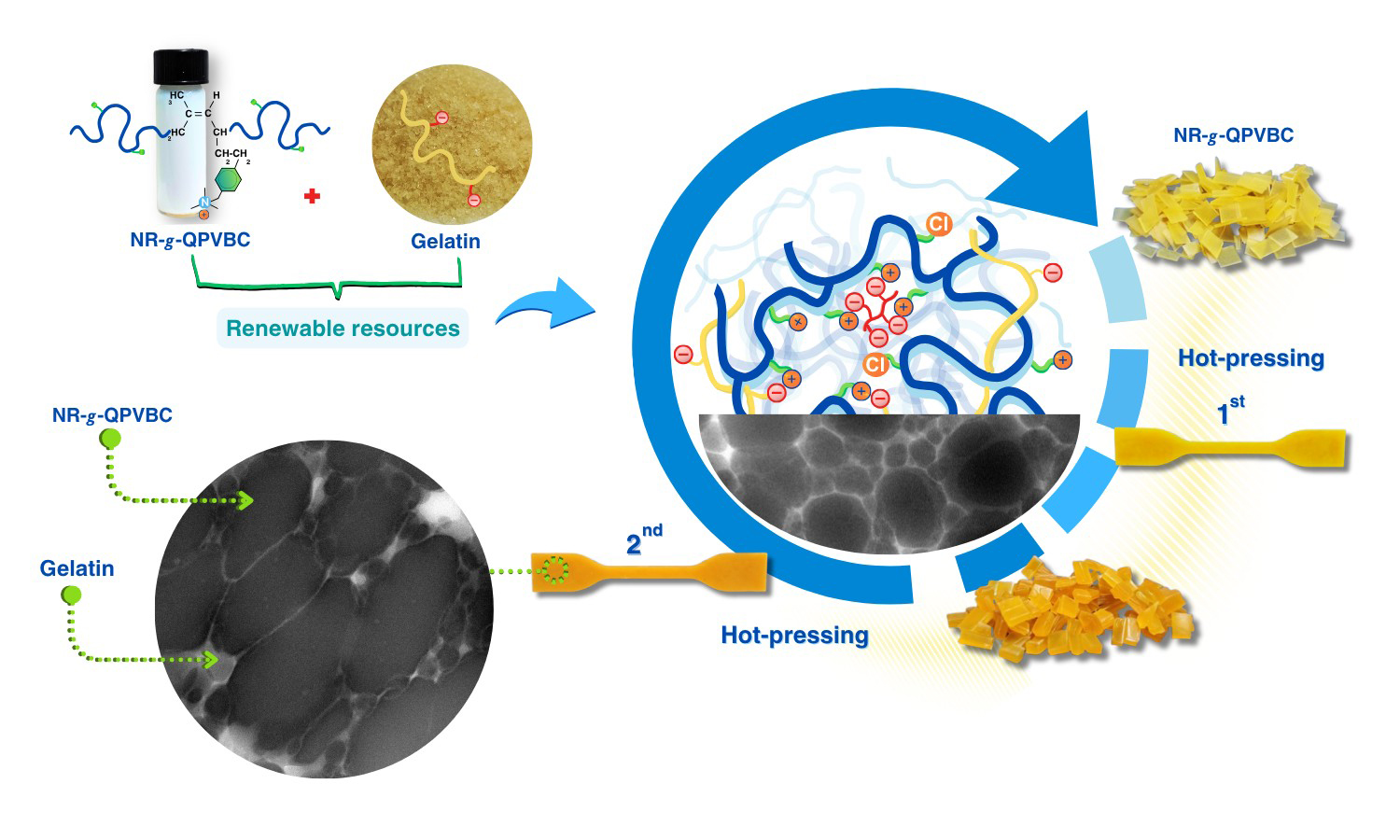
The present study has proposed a straightforward method to improve the reprocessability of modified natural rubber (NR) by blending it with gelatin (GT). The reprocessable characteristics of these blends were evaluated based on their remolding capabilities and mechanical recovery performance. In this method, poly(vinylbenzyl chloride) (PVBC) was first grafted onto NR chains to create graft copolymers known as NR-g-PVBC. The benzyl chloride groups in the graft copolymers were subsequently converted into quaternary ammonium groups, referred to as NR-g-QPVBC. This modification enabled ionic crosslinking when NR-g-QPVBC reacted with ethylenediamine tetraacetic acid. Blends were created by incorporating GT powder into the NR-g-QPVBC latex. The optimal loading level of GT was determined to be 30 wt%, as the resulting film exhibited the highest recovery of tensile properties. Initially, the film's tensile strength was measured at 15 MPa. After being remolded at 160 °C, the tensile strength decreased to 9.3 MPa, resulting in a recovery rate of 60.7% and withstanding a tensile strain of 144%. Although the NR-g-QPVBC/GT films could be remolded, their tensile properties declined with increasing remolding cycles. Therefore, this work demonstrated a practical method for producing NR-based films that could be reshaped through hot-pressing after being formed into products, increasing their reusability.
Kinsuk Naskar
Vol. 19., No.10., Pages 977-978, 2025
DOI: 10.3144/expresspolymlett.2025.73
Vol. 19., No.10., Pages 977-978, 2025
DOI: 10.3144/expresspolymlett.2025.73

This is an editorial article. It has no abstract.
Azizon Kaesaman, Tassaneeya Khunrang, Charoen Nakason
Vol. 19., No.8., Pages 753-772, 2025
DOI: 10.3144/expresspolymlett.2025.58
Vol. 19., No.8., Pages 753-772, 2025
DOI: 10.3144/expresspolymlett.2025.58
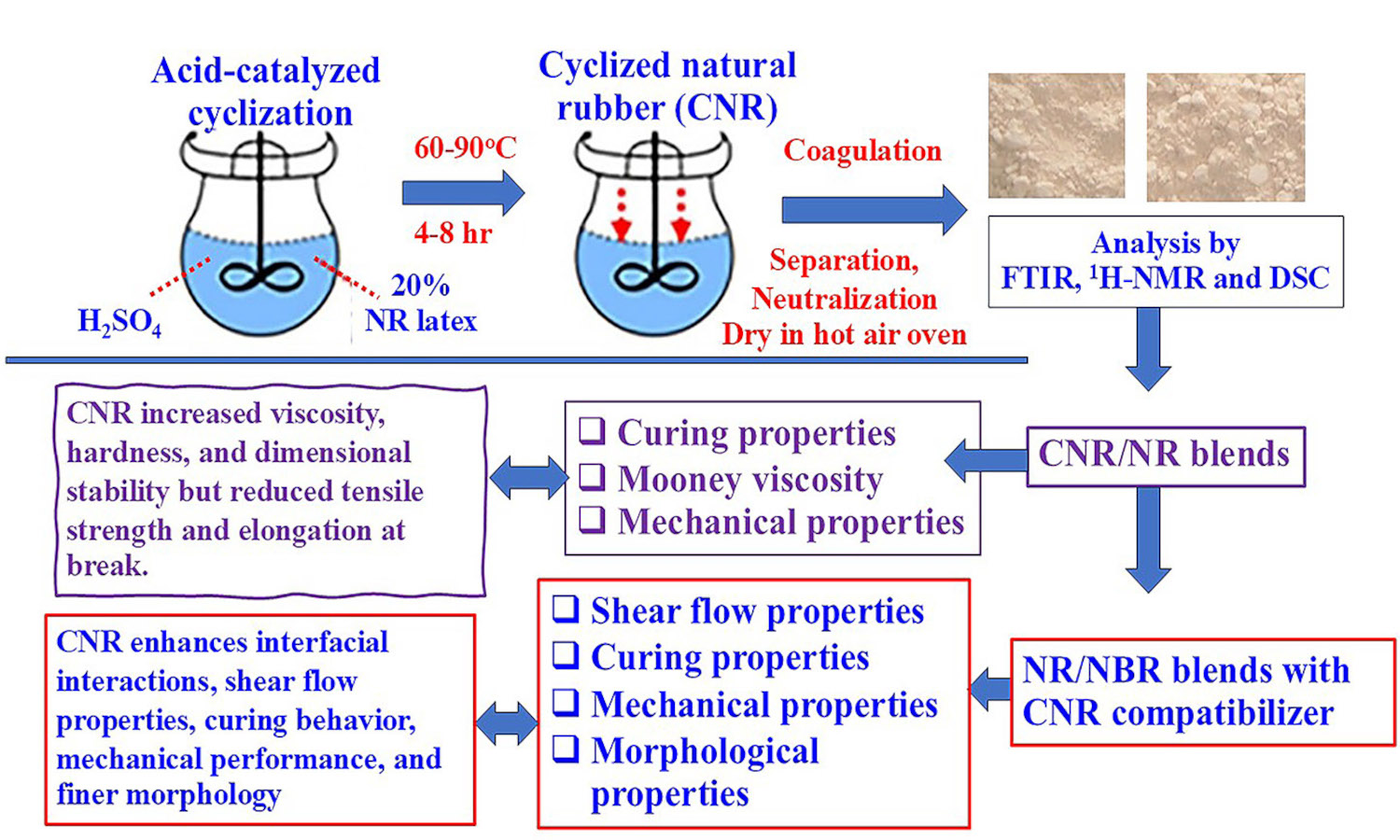
Cyclized natural rubber (CNR) was synthesized through the acid-catalyzed reaction of natural rubber (NR) latex using sulfuric acid as a catalyst and stabilized with a non-ionic surfactant. Cyclization was evaluated by iodine numbers under varying reaction times, temperatures, and NR-to-acid ratios. Fourier transform infrared spectroscopy (FTIR) and proton nuclear magnetic resonance spectroscopy (1H-NMR) confirmed the formation of cyclic structures in CNR molecules. Differential scanning calorimetry (DSC) showed that the glass transition temperature (Tg) of CNR increased with cyclization, indicating greater rigidity and less chain flexibility. CNR was then blended with NR and used as a compatibilizer in NR/acrylonitrile butadiene rubber (NBR)blends. It increased blend viscosity, hardness, and dimensional stability but reduced tensile strength and elongation due to its rigid cyclic domains. In NR/NBR blends, CNR outperformed a commercial homogenizer in enhancing interfacial interactions, leading to superior shear flow properties, curing behavior, and mechanical performance. This is attributed to the polar groups in CNR, which enhance intermolecular interactions and phase compatibility, resulting in finer phase morphology. This study highlights the potential of CNR as a versatile material for enhancing the performance of rubber compounds, with promising applications in advanced industrial formulations.




